Volume 28, Issue 4 (Autumn 2022)
Intern Med Today 2022, 28(4): 478-497 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Dalirani M, Gaeini A A, Kordi M. Investigating the Effect of Vitamin D and Calcium Supplementation Along With High-intensity Circuit Training on Lipid Profile and Body Fat in Overweight Elderly. Intern Med Today 2022; 28 (4) :478-497
URL: http://imtj.gmu.ac.ir/article-1-3926-en.html
URL: http://imtj.gmu.ac.ir/article-1-3926-en.html
1- Department of Exercise Physiology, Faculty of Sport Science, University of Tehran, Tehran, Iran.
2- Department of Exercise Physiology, Faculty of Sport Science, University of Tehran, Tehran, Iran. ,aagaeini@ut.ac.ir
2- Department of Exercise Physiology, Faculty of Sport Science, University of Tehran, Tehran, Iran. ,
Full-Text [PDF 6371 kb]
(732 Downloads)
| Abstract (HTML) (1626 Views)
Full-Text: (1225 Views)
Introduction
The high occurrence of excessive weight and obesity has emerged as a critical worldwide problem affecting roughly 1.5 billion individuals, accounting for approximately one-fifth of the world’s population [1]. According to a report in 2019, projections suggest that nearly half of the adult population in the United States will suffer from obesity by 2030 [2]. The global impact of excess weight, obesity, and central adiposity in the elderly has raised substantial public health concerns worldwide, given their correlation with an elevated susceptibility to diverse health conditions. These include high blood pressure, heart disease, metabolic disorders, abnormal lipid levels, musculoskeletal disorders, specific forms of cancer [3], and an accelerated onset of illness along with limitations in physical abilities [4]. Despite the findings of the World Health Organization (WHO) indicating that countries with advanced economies have a life expectancy of nearly 80 years, the onset and advancement of obesity-related health complications can initiate as soon as individuals enter their fifth decade of life and progress rapidly [5]. As a result, there is considerable scientific research focused on developing interventions to decrease the prevalence of obesity, especially among the elderly population who are particularly susceptible to developing this disease.
Regarding this disease mounting evidence indicates that there is often a concurrent presence of inadequate calcium intake, insufficient levels of vitamin D, obesity, and chronic non-communicable diseases [5]. This framework offers a practical strategy for individuals to follow. Several studies have indicated that calcium might play a part in the control of body weight, as evidenced by research showing that a diet rich in calcium can inhibit the accumulation of lipids in adipocytes and prevent weight gain when calorie intake is high [6, 7]. Moreover, the enhancement of uncoupling proteins, which leads to an upsurge in fat oxidation and thermogenesis may also play a role in the notable decrease in weight and body fat observed after the administration of calcium supplements [8]. Conversely, people with inadequate calcium levels may make poor dietary choices to fulfill their calcium requirements, leading to an increase in energy intake. A study discovered that the inclusion of a milk supplement, containing 1200 mg/day of calcium during weight loss reduced hunger and appetite, in addition to a tendency toward lower energy and fat intake [9]. Research has also noted the ability of calcium to suppress hunger sensations during short-term weight loss [10].
Furthermore, inadequate levels of vitamin D might play a role in an elevated differentiation of pre-adipocytes into adipocytes. The impact of vitamin D on energy regulation is strongly supported by research conducted on animal models that lack the vitamin D receptor [11]. Nevertheless, investigations on the impact of vitamin D on energy expenditure and fat oxidation in humans have produced inconclusive results [12, 13]. An investigation conducted by Wehr et al. showcased notable improvements in glucose metabolism, triglyceride levels, estrogen levels, and the regularity of menstrual cycles in women who underwent consistent vitamin D administration (20000 IU) every week for a duration of 24 weeks [14]. A two-year intervention study discovered that increased consumption of calcium from dairy products along with elevated serum vitamin D levels demonstrated a correlation with greater weight loss in response to dietary changes [15]. Furthermore, additional evidence supports the possible benefits of calcium and vitamin D supplementation in promoting fat reduction [16].
Similar to the impact of vitamin D and calcium intake, exercise training is widely acknowledged as an effective approach to improving body composition in individuals who are overweight or obese [17, 18]. Consistent moderate-to-vigorous exercise training has been demonstrated to decrease the incidence of cardiovascular disease and premature death in individuals aged 65 years and older [19, 20]. Most studies indicate that engaging in substantial amounts of exercise is linked to positive alterations in body composition, including decreases in weight, body mass index (BMI), visceral fat, and serum lipids [21, 22, 23, 24]. In addition, circuit training, which focuses on developing multiple physical strength elements simultaneously for brief intervals, has gained significant attention and may be an appropriate alternative [25].
Although previous studies have shown the potential impacts of vitamin D and calcium supplementation alongside physical activity on body composition and lipid profiles, further investigation is required to understand the potential combined outcomes of these interventions. Accordingly, this study aims to examine the impacts of a combined intervention involving calcium and vitamin D supplementation, along with high-intensity circuit training, on lipid concentrations in overweight elderly individuals.
Materials and Methods
Study subjects and design
The investigation took place in Borujerd City, Iran, from April to November 2021. The sample size was calculated via the Morgan sampling table [26]. A total of 47 elderly subjects were enrolled in this randomized, double-blind, placebo-controlled clinical study via simple random sampling. The individuals were recruited for the study through various methods, including outreach to local physicians, campus flyers, and internet advertisements. The inclusion criteria for this study were individuals of both genders aged over 60 years with 30≥ BMI ≥25 kg/m2. The participants who had undergone hormone therapy consumed anti-diabetic medications, or taken weight management drugs within the preceding 6 months were excluded from the study. The study excluded individuals who had plans to follow a specific diet or physical activity program during the study period. The subjects who smoked, abused drugs, took vitamin supplements, or had a medical condition or psychological disorder that could limit their engagement in the exercise training program were excluded from the study. The participants who fulfilled the initial inclusion requirements attended a comprehensive orientation program. During this program, the participants received an extensive overview of the study’s purpose and procedures. Consent forms were obtained and the participant’s background, including health records, was documented.
The individuals were randomly allocated to one of the 4 study groups based on BMI as follows: Group 1: 1000 mg/day calcium+50 000 IU/week vitamin D (n=12); Group 2: 50 000 IU/week vitamin D+1000 mg/day calcium+high-intensity circuit training (n=12); Group 3) High-intensity circuit training+placebo (n=13); and 4) Control (n=10) for 8 weeks (Table 1).
Double-blind administration and compliance monitoring
To ensure double-blind administration, the supplements were dispensed in coded, generic containers. The subjects were asked to return the used containers after each testing phase. Additionally, their compliance with the supplementation protocol was assessed through a post-study questionnaire, which included a compliance statement. In this study, blood specimens were obtained and analyzed prior to the beginning and following the 8 weeks of interventions.
Familiarization and training protocol
Before commencing the 8-week intervention training, the participants underwent a comprehensive familiarization session. During this session, the participants were acquainted with the testing equipment and received detailed instructions on the proper technique for all exercises. The exercise program in this study included the following 12 different types of exercises: squats, static lunges, shoulder bridge, standing single-leg calf raises, push-ups, incline bench push-ups, triceps dips, crunches, side crunches, back raises, step-ups, and side-to-side jumps over skipping rope.
During the first 2 weeks (weeks 1-2), the participants completed one circuit per session, while during weeks 3-4, they completed 2 circuits per session. Meanwhile, during weeks 5-8, they completed 3 circuits per session. The rest periods between exercises (stations) were kept minimal, allowing participants only the necessary time to transit from one station to the next (<15 s). Additionally, the inter-circuit rest period consisted of 3 min of active recovery. The participants performed all exercises using only their body weight. The target for each exercise was to complete 15-20 repetitions. In cases where the participants were unable to perform 15 or 20 repetitions continuously during an exercise, they were granted a short rest period. Following the brief pause, they were encouraged to resume the exercise and complete the remaining repetitions. The high-intensity circuit training program lasted for 8 weeks, with participants exercising 3 times per week. Every session commenced with a 5-min warm-up phase and concluded with a 5-min cool-down period. The control group participants were given instructions to maintain their regular daily activities throughout the study period without engaging in any specific exercise program [27].
The effervescent calcium tablet and its placebo with completely similar coatings and the same weight containing lactose and oral paraffin by Exir Pharma (Broujerd, Iran) and vitamin D supplement and its placebo which consisted of corn starch and Avicel and utterly similar to vitamin D by Exir Pharma Company (Boroujerd, Iran) was administered. During the research period, the study group with supplements received 1000 mg of effervescent calcium tablets at night and 50000 international units of vitamin D per week with food at noon. The supplement+high-intensity circuit training group performed the same method. In addition, they also did 3 sessions of high-intensity circuit training per week. The control group did not undergo any supplementation and refrained from engaging in any form of exercise training for the entire duration of the study, and together with the supplement and supplement+high-intensity circuit training group, fasting blood specimens were obtained from the participants 48 h before and after taking the supplement and doing the exercise training.
Anthropometric data and blood sample analyses
To assess the body weight and BMI, a body composition analyzer (InBody 570, Korea) was utilized. Height measurements were obtained using a portable stadiometer (InBody, InLab S50, Korea).
Fasting blood specimens were obtained from the participants at the Dr. Hassani Goodarzi Pathobiology Laboratory in Borujerd City, Iran, during the early morning hours following an overnight fast. To ensure prompt processing, the collected specimens were promptly centrifuged at 4°C for 10 min at a force of 2000 g. The resulting plasma and serum were stored in liquid nitrogen for further analysis. Low-density lipoprotein cholesterol (LDL-C), total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), and triglyceride (TC) levels were measured using an automated analyzer (CobasC111; Roche Diagnostics, Indianapolis, IN, USA) [28].
Statistical analysis
The Shapiro-Wilk test was utilized to evaluate the normality of the data distribution. To compare the mean differences in BMI, body fat percentage, and lipid profile between the groups, the dependent t-test, one-way analysis of variance (ANOVA), and the Tukey post hoc test were utilized. A significance level of P≤0.05 was applied to determine the statistical significance in all conducted tests. For statistical analyses, IBM Statistics SPSS software, Version 21 (SPSS Inc., Chicago, IL, USA) was utilized.
Results
Baseline characteristics
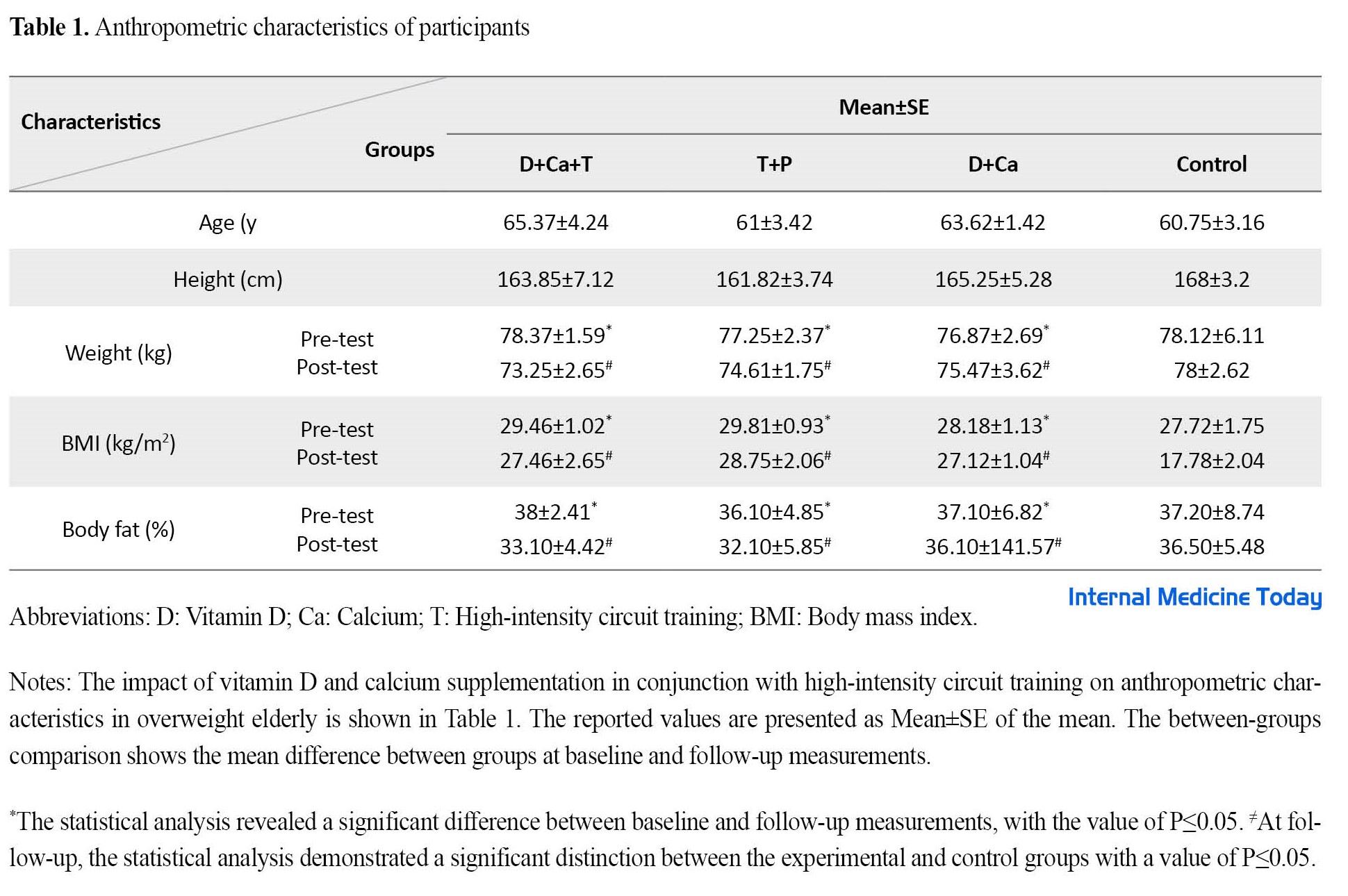
The baseline characteristics of the study participants, including demographic information, are presented in Table 1. No statistically significant differences were found among the 4 study groups in any of the assessed baseline characteristics. Statistically significant differences in BMI and body fat percentage were observed among the groups (P≤0.05), with the D+Ca+T group showing the greatest decrease. According to the results of the Tukey test, significant differences in BMI and body fat percentage were observed at the follow-up between the D+Ca+T, T+P, and D+Ca groups when compared to the control group (P≤0.05). Furthermore, Table 1 demonstrates a significant reduction in BMI and body fat percentage from pre-test to post-test in the D+Ca+T, T+P, and D+Ca groups (P≤0.05).
Profile lipid status
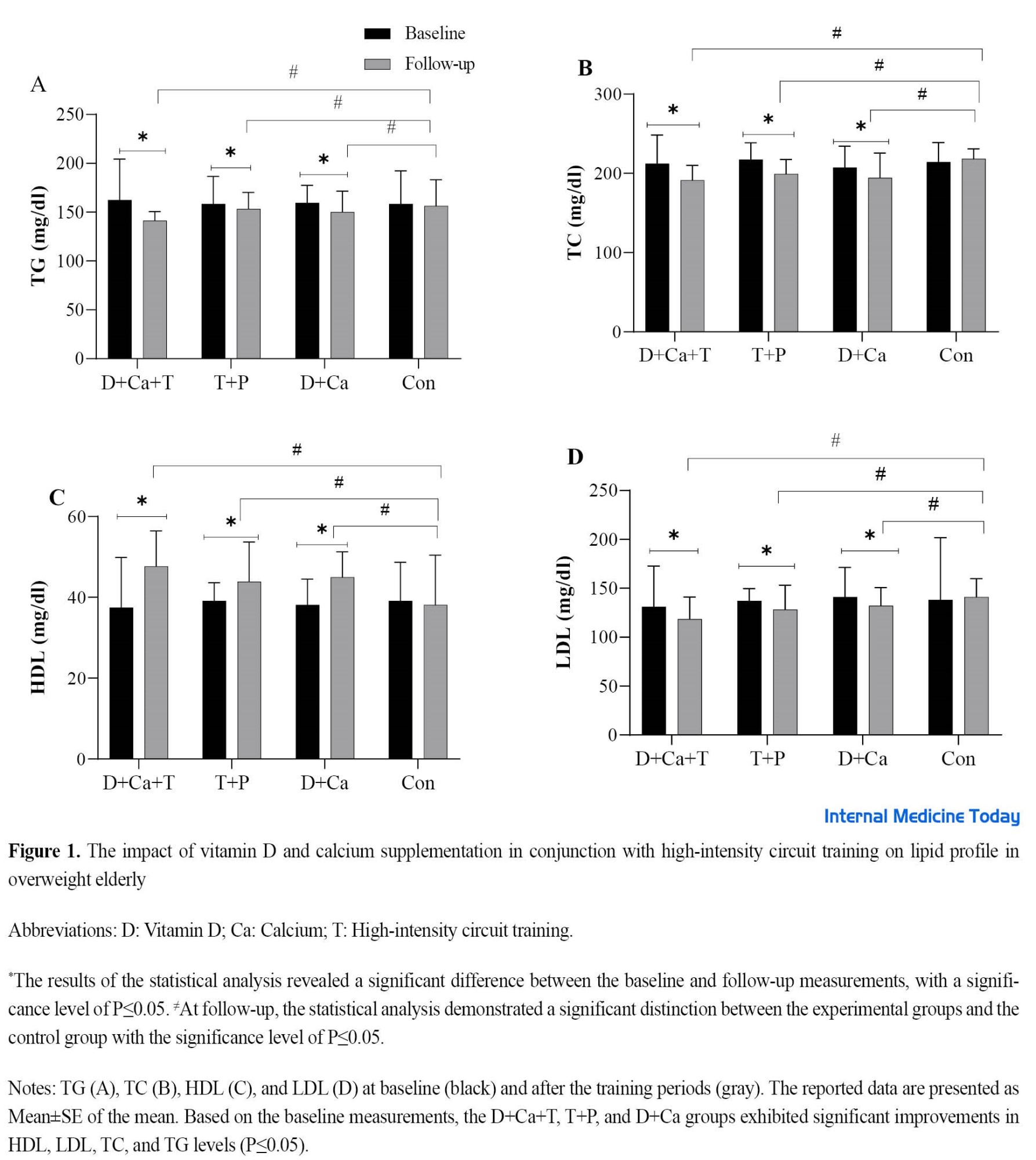
Significant differences (P≤0.05) were observed in plasma HDL, LDL, TC, and TG levels between pre-test and post-test values within each group, regardless of group type. Statistically significant differences between the groups were observed concerning HDL, LDL, TC, and TG levels, with a significance level of P≤0.05. Among the groups, the D+Ca+T combination group demonstrated the most pronounced alterations in plasma HDL, LDL, TC, and TG levels. The analysis using the Tukey test revealed significant variations in HDL, LDL, TC, and TG levels between the D+Ca+T, T+P, and D+Ca groups compared to the control group, with a significance level of P≤0.05. Statistically significant alterations in plasma HDL, LDL, TC, and TG levels were noted in the T+P and D +C groups when compared to the D+Ca+T group with a significance level of P≤0.05. Additionally, the T +P group exhibited significant improvements in HDL, LDL, TC, and TG levels compared to the D+Ca group (P≤0.05). Statistical analysis using the t tests revealed a notable rise in HDL levels from pre-test to post-test (P≤0.05) in the D+Ca+T, T+P, and D+Ca groups. Conversely, LDL, TG, and TC levels exhibited a significant reduction (P≤0.05) in the aforementioned 3 groups, while demonstrating no significant change in the control group (P≥0.05) (Figure 1).
Discussion
This study aimed to evaluate the effects of a combined intervention involving vitamin D and calcium supplementation and high-intensity circuit training on the lipid profile and body fat of overweight elderly. Significant differences were observed between the groups of D+Ca+T, T+P, and D+Ca when compared to the control group regarding LDL, HDL, TC, TG, and body fat percentage (P≤0.05), which was the main outcome of this study. The D+Ca+T, T+P, and D+Ca groups showed significant improvements in LDL, HDL, TC, TG, and body fat percentage from the pret-est to the post-test (P≤0.05). These findings suggest that regular exercise training, particularly at high intensity, combined with vitamin D and calcium supplementation, may have potential benefits for enhancing the lipid profile and reducing body fat in overweight elderly.
Extensive research has been conducted on the influence of calcium and vitamin D intake on body composition. An up-to-date meta-analysis demonstrated a notable negative correlation between calcium consumption and BMI. The analysis indicates that an incremental daily intake of 800 mg of calcium may result in a decrease of 1.1 kg/m2 in BMI [29]. Furthermore, research has demonstrated that calcium supplementation can facilitate weight loss when combined with a reduced-calorie diet and can also prevent weight increase if incorporated into a diet without calorie restrictions [7, 30]. Riedt and colleagues (2007) noted that throughout a weight reduction program, an increased calcium intake of 1800 mg/day was more effective in promoting weight loss over time compared to a moderate calcium intake of 1100 mg/day [31]. A considerable amount of evidence investigates the potential connection of vitamin D levels with various measures of adiposity, demonstrating a substantial negative link of vitamin D with total body fat [16, 25, 32]. In an investigation conducted by Vimaleswaran et al., the relationship between genetic variations, levels of vitamin D in the blood, and BMI was explored by analyzing the data of 42024 participants. The results indicated an inverse correlation between BMI and serum vitamin D levels [33]. An investigation conducted by Major et al. demonstrated that the combined intake of calcium and vitamin D led to a notable reduction in body fat among individuals having low calcium intake [16]. Furthermore, Shahar et al. [15] conducted a study indicating that individuals with enhanced calcium consumption and higher blood vitamin D levels were more successful in achieving reduced body weight through dietary interventions over 2 years [15].
There has been limited research on the combined effects of exercise, nutritional supplementation, and dieting on changes in body composition among overweight elderly individuals. An investigation conducted by Wagner et al. examined the impacts of a 12-week dietary program in premenopausal women, which included a-500 kcal restriction exercise 3 times per week, and 800 mg/day of additional calcium intake. The findings indicated that the experimental groups exhibited more substantial weight loss compared to the placebo group [34]. Furthermore, in an investigation by Babaei et al. using animal models, a combination of vitamin D supplementation and aerobic training resulted in significant reductions in overall weight, intra-abdominal fat, BMI, and caloric consumption [35]. Our findings indicated that the concurrent administration of vitamin D and calcium along with high-intensity circuit training led to improvements in adiposity and BMI. However, not all studies have consistently reported these benefits. In an investigation conducted by Holecki et al., the effects of calcium and vitamin D supplementation in conjunction with a 3-month calorie-restricted diet were examined among obese females. The findings indicated no statistically significant additional effects on body weight or reduction of fat stemming from the supplementation [36].
Furthermore, Kerksick et al. observed that at the specified amounts of 800 mg/day of calcium and 800 mg/day of calcium combined with 400 IU/day of vitamin D, there were no additional benefits of the administration of calcium and vitamin D concerning the effects of dietary and exercise interventions on body composition [5]. The differences observed in the effectiveness of weight reduction documented in various studies may be attributed to several factors, including the type of vitamin D and calcium supplementation employed, the duration of the intervention, and the incorporation of exercise. As a result, additional research is warranted to explore the impacts of varying doses of vitamin D and calcium supplementation on body weight reduction and alterations in body composition, considering diverse dietary patterns and exercise routines.
Moreover, our study revealed significant findings indicating that the concurrent implementation of high-intensity circuit training, along with calcium and vitamin D supplementation resulted in a noteworthy decrease in serum levels of TC, TG, and LDL, accompanied by an elevation in HDL among overweight elderly in the D+Ca+T, T+P, and D+Ca groups. There is a dearth of studies investigating the impacts of combining calcium-vitamin D supplementation with exercise training on lipid profiles. Consistent with the outcomes of our study, a prior investigation documented a notable decrease in serum TG levels in expectant mothers with pre-eclampsia risk who received an everyday dose of 500 mg calcium and 200 IU vitamin D for a duration of 9 weeks. However, no significant impact on other lipid profiles was observed in that study [37]. Moreover, a study conducted over 15 weeks on healthy overweight women revealed that the administration of 600 mg of calcium per day along with 200 IU of vitamin D per day resulted in a significant decrease in serum triglyceride levels [38]. According to the results of a study conducted by Asemi et al., it was determined that 8 weeks of calcium and vitamin D supplementation exhibited favorable impacts on the serum levels of triglycerides and VLDL-cholesterol in women who had a deficiency of vitamin D [39].
Following a 12-week intervention that encompassed both vitamin D supplementation and circuit training, Kim et al. observed significant improvements in abdominal fat distribution and blood lipid profiles in older women diagnosed with Type 2 diabetes and exhibiting vitamin D deficiency [25]. In our study, significant enhancements in blood lipid profiles were observed in the D+Ca+T, T+P, and D+Ca intervention groups, with the most notable improvements observed in the 2 groups that participated in circuit training. Additionally, Sung and Bae conducted a study demonstrating that older men with diabetes who engaged in walking exercise for 50 min per day, 3 times a week, at an intensity of 65%-75% of their HRmax over 24 weeks experienced significant reductions in TC and TG levels [40]. Furthermore, a study conducted by Greene et al. revealed that individuals of middle age and with obesity who engaged in endurance exercise three times per week for 12 weeks at 70% of their maximum oxygen consumption experienced noteworthy enhancements in their HDL-C levels and significant decreases in their LDL-C levels [41].
The potential positive impacts of calcium and vitamin D supplementation on lipid profile and body fat can be attributed to several mechanisms. Calcium has been demonstrated to regulate adipogenesis, influence fat metabolism by reducing lipogenesis and increasing lipolysis, stimulate adipocyte proliferation and or apoptosis, enhance thermogenesis by activating brown adipose tissue (BAT), and induce browning of white adipose tissue (WAT), inhibit dietary fat assimilation, and promote the excretion of fat in the stool via the creation of insoluble calcium-fatty complexes in the gastrointestinal tract. Additionally, calcium may also alter the structure and diversity of the intestinal microbiota, further contributing to its potential anti-obesity effects [42]. Emerging evidence suggests that regulating adipocyte number through the stimulation of apoptotic cell death may be a viable approach for preventing and treating obesity, and calcium has been implicated in this process [43]. Studies have demonstrated that elevated intracellular Ca2+ levels can induce apoptotic cell death, and this mechanism is applicable to mature adipocytes [44]. In an investigation, it was discovered that mice with diet-induced obesity exhibited a remarkable decrease in adiposity upon exposure to an amalgamation of elevated vitamin D levels and increased calcium intake. This remarkable phenomenon was attributed to the activation of the apoptotic pathway mediated by Ca2+ within their adipose tissue, providing compelling evidence for the potential regulatory role of this pathway in adiposity control [45]. Numerous scientific investigations have substantiated the pivotal role of sarco/endoplasmic reticulum Ca2+-ATPase 2b (SERCA2b)-mediated calcium cycling in the regulation of thermogenesis specifically within beige adipocytes. These findings strongly suggest an intriguing correlation between calcium signaling and the intricate process of WAT browning [ 46]. Furthermore, emerging evidence suggests that the heightened thermogenic potential of brown adipocytes, induced by calcium signaling, can be attributed to the stimulation of mitochondrial fusion and the establishment of intricate contacts between mitochondria and the endoplasmic reticulum [47]. Additionally, it has been demonstrated that heightened intracellular calcium concentrations in the liver lead to the activation of microsomal triglyceride transfer protein. This crucial protein plays a significant role in the production and secretion of VLDL, ultimately leading to a notable decrease in serum triglyceride and VLDL-cholesterol levels [48]. Furthermore, scientific investigations have revealed that the signaling pathway of vitamin D exhibits favorable impacts on the expression of genes linked to lipolysis and thermogenesis in adipose tissue [5]. Research has elucidated the multifaceted involvement of 1,25(OH)2D3 in the control of adipocyte apoptosis and differentiation. Studies have demonstrated the suppressive effects of 1,25(OH)2D3 on adipogenesis in 3T3-L1 cells, effectively impeding their transformation into fully mature adipocytes [49]. This intricate mechanism entails the stimulation of PPARγ expression while concurrently suppressing the transcriptional regulator known as CCAAT-enhancer-binding protein [50].
Our research presents both limitations and notable strengths. Firstly, our study did not explore the extended-term implications of administering calcium and vitamin D supplements, which could yield more profound outcomes. Additionally, the limited sample size utilized in our investigation may have constrained our ability to detect subtle distinctions between groups. Nevertheless, further inquiry is imperative to unravel the precise mechanisms through which vitamin D and calcium influence insulin resistance in individuals who are overweight or obese.
Conclusion
Our findings underscore the significant impacts of combining vitamin D and calcium supplementation with high-intensity circuit training on body composition and lipid profile improvement in overweight elderly. Existing evidence indicates a direct association between enhanced calcium intake and elevated levels of fat oxidation, while the signaling pathway of vitamin D has been linked to heightened thermogenic activity.
Consent to participate
All participants in the study provided informed consent before their involvement.
Ethical Considerations
Compliance with ethical guidelines
All experiments on human participants were in accordance with the ethical guidelines of the Ethics Committee of the Sport Sciences Research Institute (Code: IR.SSRI.REC.2109.1274) and the 1964 Helsinki Declaration.
Funding
This article was extracted from the master’s thesis of Maryam Dalirani approved by the Department of Exercise Physiology, University of Tehran. This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Authors' contributions
Study design and final approval: All authors; Data collection: Maryam Dalirani and Mohammadreza Kordi; Writing: Maryam Dalirani; Review and editing: Abbas Ali Gaeini and Mohammadreza Kordi.
Conflicts of interest
The authors declared no conflicts of interest.
Acknowledgments
The authors would like to thank all participants in this study for their cooperation.
References
The high occurrence of excessive weight and obesity has emerged as a critical worldwide problem affecting roughly 1.5 billion individuals, accounting for approximately one-fifth of the world’s population [1]. According to a report in 2019, projections suggest that nearly half of the adult population in the United States will suffer from obesity by 2030 [2]. The global impact of excess weight, obesity, and central adiposity in the elderly has raised substantial public health concerns worldwide, given their correlation with an elevated susceptibility to diverse health conditions. These include high blood pressure, heart disease, metabolic disorders, abnormal lipid levels, musculoskeletal disorders, specific forms of cancer [3], and an accelerated onset of illness along with limitations in physical abilities [4]. Despite the findings of the World Health Organization (WHO) indicating that countries with advanced economies have a life expectancy of nearly 80 years, the onset and advancement of obesity-related health complications can initiate as soon as individuals enter their fifth decade of life and progress rapidly [5]. As a result, there is considerable scientific research focused on developing interventions to decrease the prevalence of obesity, especially among the elderly population who are particularly susceptible to developing this disease.
Regarding this disease mounting evidence indicates that there is often a concurrent presence of inadequate calcium intake, insufficient levels of vitamin D, obesity, and chronic non-communicable diseases [5]. This framework offers a practical strategy for individuals to follow. Several studies have indicated that calcium might play a part in the control of body weight, as evidenced by research showing that a diet rich in calcium can inhibit the accumulation of lipids in adipocytes and prevent weight gain when calorie intake is high [6, 7]. Moreover, the enhancement of uncoupling proteins, which leads to an upsurge in fat oxidation and thermogenesis may also play a role in the notable decrease in weight and body fat observed after the administration of calcium supplements [8]. Conversely, people with inadequate calcium levels may make poor dietary choices to fulfill their calcium requirements, leading to an increase in energy intake. A study discovered that the inclusion of a milk supplement, containing 1200 mg/day of calcium during weight loss reduced hunger and appetite, in addition to a tendency toward lower energy and fat intake [9]. Research has also noted the ability of calcium to suppress hunger sensations during short-term weight loss [10].
Furthermore, inadequate levels of vitamin D might play a role in an elevated differentiation of pre-adipocytes into adipocytes. The impact of vitamin D on energy regulation is strongly supported by research conducted on animal models that lack the vitamin D receptor [11]. Nevertheless, investigations on the impact of vitamin D on energy expenditure and fat oxidation in humans have produced inconclusive results [12, 13]. An investigation conducted by Wehr et al. showcased notable improvements in glucose metabolism, triglyceride levels, estrogen levels, and the regularity of menstrual cycles in women who underwent consistent vitamin D administration (20000 IU) every week for a duration of 24 weeks [14]. A two-year intervention study discovered that increased consumption of calcium from dairy products along with elevated serum vitamin D levels demonstrated a correlation with greater weight loss in response to dietary changes [15]. Furthermore, additional evidence supports the possible benefits of calcium and vitamin D supplementation in promoting fat reduction [16].
Similar to the impact of vitamin D and calcium intake, exercise training is widely acknowledged as an effective approach to improving body composition in individuals who are overweight or obese [17, 18]. Consistent moderate-to-vigorous exercise training has been demonstrated to decrease the incidence of cardiovascular disease and premature death in individuals aged 65 years and older [19, 20]. Most studies indicate that engaging in substantial amounts of exercise is linked to positive alterations in body composition, including decreases in weight, body mass index (BMI), visceral fat, and serum lipids [21, 22, 23, 24]. In addition, circuit training, which focuses on developing multiple physical strength elements simultaneously for brief intervals, has gained significant attention and may be an appropriate alternative [25].
Although previous studies have shown the potential impacts of vitamin D and calcium supplementation alongside physical activity on body composition and lipid profiles, further investigation is required to understand the potential combined outcomes of these interventions. Accordingly, this study aims to examine the impacts of a combined intervention involving calcium and vitamin D supplementation, along with high-intensity circuit training, on lipid concentrations in overweight elderly individuals.
Materials and Methods
Study subjects and design
The investigation took place in Borujerd City, Iran, from April to November 2021. The sample size was calculated via the Morgan sampling table [26]. A total of 47 elderly subjects were enrolled in this randomized, double-blind, placebo-controlled clinical study via simple random sampling. The individuals were recruited for the study through various methods, including outreach to local physicians, campus flyers, and internet advertisements. The inclusion criteria for this study were individuals of both genders aged over 60 years with 30≥ BMI ≥25 kg/m2. The participants who had undergone hormone therapy consumed anti-diabetic medications, or taken weight management drugs within the preceding 6 months were excluded from the study. The study excluded individuals who had plans to follow a specific diet or physical activity program during the study period. The subjects who smoked, abused drugs, took vitamin supplements, or had a medical condition or psychological disorder that could limit their engagement in the exercise training program were excluded from the study. The participants who fulfilled the initial inclusion requirements attended a comprehensive orientation program. During this program, the participants received an extensive overview of the study’s purpose and procedures. Consent forms were obtained and the participant’s background, including health records, was documented.
The individuals were randomly allocated to one of the 4 study groups based on BMI as follows: Group 1: 1000 mg/day calcium+50 000 IU/week vitamin D (n=12); Group 2: 50 000 IU/week vitamin D+1000 mg/day calcium+high-intensity circuit training (n=12); Group 3) High-intensity circuit training+placebo (n=13); and 4) Control (n=10) for 8 weeks (Table 1).

Double-blind administration and compliance monitoring
To ensure double-blind administration, the supplements were dispensed in coded, generic containers. The subjects were asked to return the used containers after each testing phase. Additionally, their compliance with the supplementation protocol was assessed through a post-study questionnaire, which included a compliance statement. In this study, blood specimens were obtained and analyzed prior to the beginning and following the 8 weeks of interventions.
Familiarization and training protocol
Before commencing the 8-week intervention training, the participants underwent a comprehensive familiarization session. During this session, the participants were acquainted with the testing equipment and received detailed instructions on the proper technique for all exercises. The exercise program in this study included the following 12 different types of exercises: squats, static lunges, shoulder bridge, standing single-leg calf raises, push-ups, incline bench push-ups, triceps dips, crunches, side crunches, back raises, step-ups, and side-to-side jumps over skipping rope.
During the first 2 weeks (weeks 1-2), the participants completed one circuit per session, while during weeks 3-4, they completed 2 circuits per session. Meanwhile, during weeks 5-8, they completed 3 circuits per session. The rest periods between exercises (stations) were kept minimal, allowing participants only the necessary time to transit from one station to the next (<15 s). Additionally, the inter-circuit rest period consisted of 3 min of active recovery. The participants performed all exercises using only their body weight. The target for each exercise was to complete 15-20 repetitions. In cases where the participants were unable to perform 15 or 20 repetitions continuously during an exercise, they were granted a short rest period. Following the brief pause, they were encouraged to resume the exercise and complete the remaining repetitions. The high-intensity circuit training program lasted for 8 weeks, with participants exercising 3 times per week. Every session commenced with a 5-min warm-up phase and concluded with a 5-min cool-down period. The control group participants were given instructions to maintain their regular daily activities throughout the study period without engaging in any specific exercise program [27].
The effervescent calcium tablet and its placebo with completely similar coatings and the same weight containing lactose and oral paraffin by Exir Pharma (Broujerd, Iran) and vitamin D supplement and its placebo which consisted of corn starch and Avicel and utterly similar to vitamin D by Exir Pharma Company (Boroujerd, Iran) was administered. During the research period, the study group with supplements received 1000 mg of effervescent calcium tablets at night and 50000 international units of vitamin D per week with food at noon. The supplement+high-intensity circuit training group performed the same method. In addition, they also did 3 sessions of high-intensity circuit training per week. The control group did not undergo any supplementation and refrained from engaging in any form of exercise training for the entire duration of the study, and together with the supplement and supplement+high-intensity circuit training group, fasting blood specimens were obtained from the participants 48 h before and after taking the supplement and doing the exercise training.
Anthropometric data and blood sample analyses
To assess the body weight and BMI, a body composition analyzer (InBody 570, Korea) was utilized. Height measurements were obtained using a portable stadiometer (InBody, InLab S50, Korea).
Fasting blood specimens were obtained from the participants at the Dr. Hassani Goodarzi Pathobiology Laboratory in Borujerd City, Iran, during the early morning hours following an overnight fast. To ensure prompt processing, the collected specimens were promptly centrifuged at 4°C for 10 min at a force of 2000 g. The resulting plasma and serum were stored in liquid nitrogen for further analysis. Low-density lipoprotein cholesterol (LDL-C), total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), and triglyceride (TC) levels were measured using an automated analyzer (CobasC111; Roche Diagnostics, Indianapolis, IN, USA) [28].
Statistical analysis
The Shapiro-Wilk test was utilized to evaluate the normality of the data distribution. To compare the mean differences in BMI, body fat percentage, and lipid profile between the groups, the dependent t-test, one-way analysis of variance (ANOVA), and the Tukey post hoc test were utilized. A significance level of P≤0.05 was applied to determine the statistical significance in all conducted tests. For statistical analyses, IBM Statistics SPSS software, Version 21 (SPSS Inc., Chicago, IL, USA) was utilized.
Results
Baseline characteristics
The baseline characteristics of the study participants, including demographic information, are presented in Table 1. No statistically significant differences were found among the 4 study groups in any of the assessed baseline characteristics. Statistically significant differences in BMI and body fat percentage were observed among the groups (P≤0.05), with the D+Ca+T group showing the greatest decrease. According to the results of the Tukey test, significant differences in BMI and body fat percentage were observed at the follow-up between the D+Ca+T, T+P, and D+Ca groups when compared to the control group (P≤0.05). Furthermore, Table 1 demonstrates a significant reduction in BMI and body fat percentage from pre-test to post-test in the D+Ca+T, T+P, and D+Ca groups (P≤0.05).
Profile lipid status
Significant differences (P≤0.05) were observed in plasma HDL, LDL, TC, and TG levels between pre-test and post-test values within each group, regardless of group type. Statistically significant differences between the groups were observed concerning HDL, LDL, TC, and TG levels, with a significance level of P≤0.05. Among the groups, the D+Ca+T combination group demonstrated the most pronounced alterations in plasma HDL, LDL, TC, and TG levels. The analysis using the Tukey test revealed significant variations in HDL, LDL, TC, and TG levels between the D+Ca+T, T+P, and D+Ca groups compared to the control group, with a significance level of P≤0.05. Statistically significant alterations in plasma HDL, LDL, TC, and TG levels were noted in the T+P and D +C groups when compared to the D+Ca+T group with a significance level of P≤0.05. Additionally, the T +P group exhibited significant improvements in HDL, LDL, TC, and TG levels compared to the D+Ca group (P≤0.05). Statistical analysis using the t tests revealed a notable rise in HDL levels from pre-test to post-test (P≤0.05) in the D+Ca+T, T+P, and D+Ca groups. Conversely, LDL, TG, and TC levels exhibited a significant reduction (P≤0.05) in the aforementioned 3 groups, while demonstrating no significant change in the control group (P≥0.05) (Figure 1).
Discussion
This study aimed to evaluate the effects of a combined intervention involving vitamin D and calcium supplementation and high-intensity circuit training on the lipid profile and body fat of overweight elderly. Significant differences were observed between the groups of D+Ca+T, T+P, and D+Ca when compared to the control group regarding LDL, HDL, TC, TG, and body fat percentage (P≤0.05), which was the main outcome of this study. The D+Ca+T, T+P, and D+Ca groups showed significant improvements in LDL, HDL, TC, TG, and body fat percentage from the pret-est to the post-test (P≤0.05). These findings suggest that regular exercise training, particularly at high intensity, combined with vitamin D and calcium supplementation, may have potential benefits for enhancing the lipid profile and reducing body fat in overweight elderly.
Extensive research has been conducted on the influence of calcium and vitamin D intake on body composition. An up-to-date meta-analysis demonstrated a notable negative correlation between calcium consumption and BMI. The analysis indicates that an incremental daily intake of 800 mg of calcium may result in a decrease of 1.1 kg/m2 in BMI [29]. Furthermore, research has demonstrated that calcium supplementation can facilitate weight loss when combined with a reduced-calorie diet and can also prevent weight increase if incorporated into a diet without calorie restrictions [7, 30]. Riedt and colleagues (2007) noted that throughout a weight reduction program, an increased calcium intake of 1800 mg/day was more effective in promoting weight loss over time compared to a moderate calcium intake of 1100 mg/day [31]. A considerable amount of evidence investigates the potential connection of vitamin D levels with various measures of adiposity, demonstrating a substantial negative link of vitamin D with total body fat [16, 25, 32]. In an investigation conducted by Vimaleswaran et al., the relationship between genetic variations, levels of vitamin D in the blood, and BMI was explored by analyzing the data of 42024 participants. The results indicated an inverse correlation between BMI and serum vitamin D levels [33]. An investigation conducted by Major et al. demonstrated that the combined intake of calcium and vitamin D led to a notable reduction in body fat among individuals having low calcium intake [16]. Furthermore, Shahar et al. [15] conducted a study indicating that individuals with enhanced calcium consumption and higher blood vitamin D levels were more successful in achieving reduced body weight through dietary interventions over 2 years [15].
There has been limited research on the combined effects of exercise, nutritional supplementation, and dieting on changes in body composition among overweight elderly individuals. An investigation conducted by Wagner et al. examined the impacts of a 12-week dietary program in premenopausal women, which included a-500 kcal restriction exercise 3 times per week, and 800 mg/day of additional calcium intake. The findings indicated that the experimental groups exhibited more substantial weight loss compared to the placebo group [34]. Furthermore, in an investigation by Babaei et al. using animal models, a combination of vitamin D supplementation and aerobic training resulted in significant reductions in overall weight, intra-abdominal fat, BMI, and caloric consumption [35]. Our findings indicated that the concurrent administration of vitamin D and calcium along with high-intensity circuit training led to improvements in adiposity and BMI. However, not all studies have consistently reported these benefits. In an investigation conducted by Holecki et al., the effects of calcium and vitamin D supplementation in conjunction with a 3-month calorie-restricted diet were examined among obese females. The findings indicated no statistically significant additional effects on body weight or reduction of fat stemming from the supplementation [36].
Furthermore, Kerksick et al. observed that at the specified amounts of 800 mg/day of calcium and 800 mg/day of calcium combined with 400 IU/day of vitamin D, there were no additional benefits of the administration of calcium and vitamin D concerning the effects of dietary and exercise interventions on body composition [5]. The differences observed in the effectiveness of weight reduction documented in various studies may be attributed to several factors, including the type of vitamin D and calcium supplementation employed, the duration of the intervention, and the incorporation of exercise. As a result, additional research is warranted to explore the impacts of varying doses of vitamin D and calcium supplementation on body weight reduction and alterations in body composition, considering diverse dietary patterns and exercise routines.
Moreover, our study revealed significant findings indicating that the concurrent implementation of high-intensity circuit training, along with calcium and vitamin D supplementation resulted in a noteworthy decrease in serum levels of TC, TG, and LDL, accompanied by an elevation in HDL among overweight elderly in the D+Ca+T, T+P, and D+Ca groups. There is a dearth of studies investigating the impacts of combining calcium-vitamin D supplementation with exercise training on lipid profiles. Consistent with the outcomes of our study, a prior investigation documented a notable decrease in serum TG levels in expectant mothers with pre-eclampsia risk who received an everyday dose of 500 mg calcium and 200 IU vitamin D for a duration of 9 weeks. However, no significant impact on other lipid profiles was observed in that study [37]. Moreover, a study conducted over 15 weeks on healthy overweight women revealed that the administration of 600 mg of calcium per day along with 200 IU of vitamin D per day resulted in a significant decrease in serum triglyceride levels [38]. According to the results of a study conducted by Asemi et al., it was determined that 8 weeks of calcium and vitamin D supplementation exhibited favorable impacts on the serum levels of triglycerides and VLDL-cholesterol in women who had a deficiency of vitamin D [39].
Following a 12-week intervention that encompassed both vitamin D supplementation and circuit training, Kim et al. observed significant improvements in abdominal fat distribution and blood lipid profiles in older women diagnosed with Type 2 diabetes and exhibiting vitamin D deficiency [25]. In our study, significant enhancements in blood lipid profiles were observed in the D+Ca+T, T+P, and D+Ca intervention groups, with the most notable improvements observed in the 2 groups that participated in circuit training. Additionally, Sung and Bae conducted a study demonstrating that older men with diabetes who engaged in walking exercise for 50 min per day, 3 times a week, at an intensity of 65%-75% of their HRmax over 24 weeks experienced significant reductions in TC and TG levels [40]. Furthermore, a study conducted by Greene et al. revealed that individuals of middle age and with obesity who engaged in endurance exercise three times per week for 12 weeks at 70% of their maximum oxygen consumption experienced noteworthy enhancements in their HDL-C levels and significant decreases in their LDL-C levels [41].
The potential positive impacts of calcium and vitamin D supplementation on lipid profile and body fat can be attributed to several mechanisms. Calcium has been demonstrated to regulate adipogenesis, influence fat metabolism by reducing lipogenesis and increasing lipolysis, stimulate adipocyte proliferation and or apoptosis, enhance thermogenesis by activating brown adipose tissue (BAT), and induce browning of white adipose tissue (WAT), inhibit dietary fat assimilation, and promote the excretion of fat in the stool via the creation of insoluble calcium-fatty complexes in the gastrointestinal tract. Additionally, calcium may also alter the structure and diversity of the intestinal microbiota, further contributing to its potential anti-obesity effects [42]. Emerging evidence suggests that regulating adipocyte number through the stimulation of apoptotic cell death may be a viable approach for preventing and treating obesity, and calcium has been implicated in this process [43]. Studies have demonstrated that elevated intracellular Ca2+ levels can induce apoptotic cell death, and this mechanism is applicable to mature adipocytes [44]. In an investigation, it was discovered that mice with diet-induced obesity exhibited a remarkable decrease in adiposity upon exposure to an amalgamation of elevated vitamin D levels and increased calcium intake. This remarkable phenomenon was attributed to the activation of the apoptotic pathway mediated by Ca2+ within their adipose tissue, providing compelling evidence for the potential regulatory role of this pathway in adiposity control [45]. Numerous scientific investigations have substantiated the pivotal role of sarco/endoplasmic reticulum Ca2+-ATPase 2b (SERCA2b)-mediated calcium cycling in the regulation of thermogenesis specifically within beige adipocytes. These findings strongly suggest an intriguing correlation between calcium signaling and the intricate process of WAT browning [ 46]. Furthermore, emerging evidence suggests that the heightened thermogenic potential of brown adipocytes, induced by calcium signaling, can be attributed to the stimulation of mitochondrial fusion and the establishment of intricate contacts between mitochondria and the endoplasmic reticulum [47]. Additionally, it has been demonstrated that heightened intracellular calcium concentrations in the liver lead to the activation of microsomal triglyceride transfer protein. This crucial protein plays a significant role in the production and secretion of VLDL, ultimately leading to a notable decrease in serum triglyceride and VLDL-cholesterol levels [48]. Furthermore, scientific investigations have revealed that the signaling pathway of vitamin D exhibits favorable impacts on the expression of genes linked to lipolysis and thermogenesis in adipose tissue [5]. Research has elucidated the multifaceted involvement of 1,25(OH)2D3 in the control of adipocyte apoptosis and differentiation. Studies have demonstrated the suppressive effects of 1,25(OH)2D3 on adipogenesis in 3T3-L1 cells, effectively impeding their transformation into fully mature adipocytes [49]. This intricate mechanism entails the stimulation of PPARγ expression while concurrently suppressing the transcriptional regulator known as CCAAT-enhancer-binding protein [50].
Our research presents both limitations and notable strengths. Firstly, our study did not explore the extended-term implications of administering calcium and vitamin D supplements, which could yield more profound outcomes. Additionally, the limited sample size utilized in our investigation may have constrained our ability to detect subtle distinctions between groups. Nevertheless, further inquiry is imperative to unravel the precise mechanisms through which vitamin D and calcium influence insulin resistance in individuals who are overweight or obese.
Conclusion
Our findings underscore the significant impacts of combining vitamin D and calcium supplementation with high-intensity circuit training on body composition and lipid profile improvement in overweight elderly. Existing evidence indicates a direct association between enhanced calcium intake and elevated levels of fat oxidation, while the signaling pathway of vitamin D has been linked to heightened thermogenic activity.
Consent to participate
All participants in the study provided informed consent before their involvement.
Ethical Considerations
Compliance with ethical guidelines
All experiments on human participants were in accordance with the ethical guidelines of the Ethics Committee of the Sport Sciences Research Institute (Code: IR.SSRI.REC.2109.1274) and the 1964 Helsinki Declaration.
Funding
This article was extracted from the master’s thesis of Maryam Dalirani approved by the Department of Exercise Physiology, University of Tehran. This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Authors' contributions
Study design and final approval: All authors; Data collection: Maryam Dalirani and Mohammadreza Kordi; Writing: Maryam Dalirani; Review and editing: Abbas Ali Gaeini and Mohammadreza Kordi.
Conflicts of interest
The authors declared no conflicts of interest.
Acknowledgments
The authors would like to thank all participants in this study for their cooperation.
References
- Popkin BM, Adair LS, Ng SW. Global nutrition transition and the pandemic of obesity in developing countries. Nutrition Reviews. 2012; 70(1):3-21. [DOI:10.1111/j.1753-4887.2011.00456.x] [PMID]
- Ward ZJ, Bleich SN, Cradock AL, Barrett JL, Giles CM, Flax C, et al. Projected U.S. state-level prevalence of adult obesity and severe obesity. New England Journal of Medicine. 2019; 381(25):2440-50. [DOI:10.1056/NEJMsa1909301] [PMID]
- Houston DK, Nicklas BJ, Zizza CA. Weighty concerns: The growing prevalence of obesity among older adults. Journal of the American Dietetic Association. 2009; 109(11):1886-95. [DOI:10.1016/j.jada.2009.08.014] [PMID]
- Ramsay SE, Whincup PH, Shaper AG, Wannamethee SG. The relations of body composition and adiposity measures to ill health and physical disability in elderly men. American Journal of Epidemiology. 2006; 164(5):459-69. [DOI:10.1093/aje/kwj217] [PMID]
- Kerksick CM, Roberts MD, Campbell BI, Galbreath MM, Taylor LW, Wilborn CD, et al. Differential impact of calcium and vitamin D on body composition changes in post-menopausal women following a restricted energy diet and exercise program. Nutrients. 2020; 12(3):713. [DOI:10.3390/nu12030713] [PMID] [PMCID]
- Shi H, Dirienzo D, Zemel MB. Effects of dietary calcium on adipocyte lipid metabolism and body weight regulation in energy-restricted aP2-agouti transgenic mice. The FASEB Journal. 2001; 15(2):291-3. [DOI:10.1096/fj.00-0584fje] [PMID]
- Teegarden D, Zemel MB. Dairy product components and weight regulation: Symposium overview. The Journal of Nutrition. 2003; 133(1):243S-4S. [DOI:10.1093/jn/133.1.243S] [PMID]
- Soares MJ, Chan She Ping-Delfos W, Ghanbari MH. Calcium and vitamin D for obesity: A review of randomized controlled trials. European Journal of Clinical Nutrition. 2011; 65(9):994-1004. [DOI:10.1038/ejcn.2011.106] [PMID]
- Gilbert JA, Joanisse DR, Chaput JP, Miegueu P, Cianflone K, Alméras N, et al. Milk supplementation facilitates appetite control in obese women during weight loss: A randomised, single-blind, placebo-controlled trial. British Journal of Nutrition. 2011; 105(1):133-43. [DOI:10.1017/S0007114510003119] [PMID]
- Kabrnová-Hlavatá K, Hainer V, Gojová M, Hlavatý P, Kopský V, Nedvídková J, et al. Calcium intake and the outcome of short-term weight management. Physiological Research. 2008; 57(2):237-45. [DOI:10.33549/physiolres.931057] [PMID]
- Wong KE, Szeto FL, Zhang W, Ye H, Kong J, Zhang Z, et al. Involvement of the vitamin D receptor in energy metabolism: Regulation of uncoupling proteins. American Journal of Physiology-Endocrinology and Metabolism. 2009; 296(4):E820-8. [DOI:10.1152/ajpendo.90763.2008] [PMID] [PMCID]
- Teegarden D, White KM, Lyle RM, Zemel MB, Van Loan MD, Matkovic V, et al. Calcium and dairy product modulation of lipid utilization and energy expenditure. Obesity. 2008; 16(7):1566-72. [DOI:10.1038/oby.2008.232] [PMID]
- Boon N, Hul GB, Sicard A, Kole E, Van Den Berg ER, Viguerie N, et al. The effects of increasing serum calcitriol on energy and fat metabolism and gene expression. Obesity. 2006; 14(10):1739-46. [DOI:10.1038/oby.2006.200] [PMID]
- Wehr E, Pieber TR, Obermayer-Pietsch B. Effect of vitamin D3 treatment on glucose metabolism and menstrual frequency in polycystic ovary syndrome women: A pilot study. Journal of Endocrinological Investigation. 2011; 34(10):757-63. [PMID]
- Shahar DR, Schwarzfuchs D, Fraser D, Vardi H, Thiery J, Fiedler GM, et al. Dairy calcium intake, serum vitamin D, and successful weight loss. The American Journal of Clinical Nutrition. 2010; 92(5):1017-22. [DOI:10.3945/ajcn.2010.29355] [PMID]
- Major GC, Alarie FP, Doré J, Tremblay A. Calcium plus vitamin D supplementation and fat mass loss in female very low-calcium consumers: Potential link with a calcium-specific appetite control. British journal of Nutrition. 2008; 101(5):659-63. [DOI:10.1017/S0007114508030808] [PMID]
- Poirier P, Després JP. Exercise in weight management of obesity. Cardiology Clinics. 2001; 19(3):459-70. [DOI:10.1016/S0733-8651(05)70229-0] [PMID]
- Johansson K, Neovius M, Hemmingsson E. Effects of anti-obesity drugs, diet, and exercise on weight-loss maintenance after a very-low-calorie diet or low-calorie diet: a systematic review and meta-analysis of randomized controlled trials. The American Journal of Clinical Nutrition. 2014; 99(1):14-23. [DOI:10.3945/ajcn.113.070052] [PMID] [PMCID]
- Hamer M, Stamatakis E. Low-dose physical activity attenuates cardiovascular disease mortality in men and women with clustered metabolic risk factors. Circulation: Cardiovascular Quality and Outcomes. 2012; 5(4):494-9. [DOI:10.1161/CIRCOUTCOMES.112.965434] [PMID]
- Middleton LE, Barnes DE, Lui LY, Yaffe K. Physical activity over the life course and its association with cognitive performance and impairment in old age. Journal of the American Geriatrics Society. 2010; 58(7):1322-6. [DOI:10.1111/j.1532-5415.2010.02903.x] [PMID] [PMCID]
- Pescatello LS, Murphy D, Costanzo D. Low-intensity physical activity benefits blood lipids and lipoproteins in older adults living at home. Age and Ageing. 2000; 29(5):433-9. [DOI:10.1093/ageing/29.5.433] [PMID]
- Rosique-Esteban N, Babio N, Díaz-López A, Romaguera D, Alfredo Martínez J, Sanchez VM, et al. Leisure-time physical activity at moderate and high intensity is associated with parameters of body composition, muscle strength and sarcopenia in aged adults with obesity and metabolic syndrome from the PREDIMED-Plus study. Clinical Nutrition. 2019; 38(3):1324-31. [DOI:10.1016/j.clnu.2018.05.023] [PMID]
- Dunsky A, Zach S, Zeev A, Goldbourt U, Shimony T, Goldsmith R, et al. Level of physical activity and anthropometric characteristics in old age-results from a national health survey. European Review of Aging and Physical Activity. 2014; 11(2):149-57. [DOI:10.1007/s11556-014-0139-y]
- Silva MR, Silva HH, Capkauskiene S, Rosado-Marques V, Machado-Rodrigues A, Nogueira H, et al. Cardiovascular and metabolic risk factors in physically active and inactive Portuguese middle-aged adults: A cross-sectional study. Science & Sports. 2019; 35(4):e91-8. [DOI:10.1016/j.scispo.2019.06.009]
- Kim HJ, Kang CK, Park H, Lee MG. Effects of vitamin D supplementation and circuit training on indices of obesity and insulin resistance in T2D and vitamin D deficient elderly women. Journal of Exercise Nutrition & Biochemistry. 2014; 18(3):249-57. [DOI:10.5717/jenb.2014.18.3.249] [PMID] [PMCID]
- Krejcie RV, Morgan DW. Determining sample size for research activities. Educational and Psychological Measurement. 1970; 30(3):607-10. [DOI:10.1177/001316447003000308]
- Vrachimis A, Hadjicharalambous M, Tyler C. The effect of circuit training on resting heart rate variability, cardiovascular disease risk factors and physical fitness in healthy untrained adults. Health. 2016; 8(2):144-55. [DOI:10.4236/health.2016.82017]
- Ghardashi-Afousi A, Davoodi M, Hesamabadi BK, Asvadi-Fard M, Bigi MAB, Izadi MR, et al. Improved carotid intima-media thickness-induced high-intensity interval training associated with decreased serum levels of Dkk-1 and sclerostin in type 2 diabetes. Journal of Diabetes and its Complications. 2020; 34(1):107469. [DOI:10.1016/j.jdiacomp.2019.107469] [PMID]
- Dougkas A, Reynolds CK, Givens ID, Elwood PC, Minihane AM. Associations between dairy consumption and body weight: A review of the evidence and underlying mechanisms. Nutrition Research Reviews. 2011; 24(1):72-95. [DOI:10.1017/S095442241000034X] [PMID]
- Lappe JM, Rafferty KA, Davies KM, Lypaczewski G. Girls on a high-calcium diet gain weight at the same rate as girls on a normal diet: A pilot study. Journal of the American Dietetic Association. 2004; 104(9):1361-7. [DOI:10.1016/j.jada.2004.06.025] [PMID]
- Riedt CS, Schlussel Y, Von Thun N, Ambia-Sobhan H, Stahl T, Field MP, et al. Premenopausal overweight women do not lose bone during moderate weight loss with adequate or higher calcium intake. The American Journal of Clinical Nutrition. 2007; 85(4):972-80. [DOI:10.1093/ajcn/85.4.972] [PMID] [PMCID]
- Freedman BI, Wagenknecht LE, Hairston KG, Bowden DW, Carr JJ, Hightower RC, et al. Vitamin D, adiposity, and calcified atherosclerotic plaque in African-Americans. The Journal of Clinical Endocrinology & Metabolism. 2010; 95(3):1076-83. [DOI:10.1210/jc.2009-1797] [PMID] [PMCID]
- Vimaleswaran KS, Berry DJ, Lu C, Tikkanen E, Pilz S, Hiraki LT, et al. Causal relationship between obesity and vitamin D status: Bi-directional Mendelian randomization analysis of multiple cohorts. PLoS Medicine. 2013; 10(2):e1001383. [PMID]
- Wagner G, Kindrick S, Hertzler S, DiSilvestro RA. Effects of various forms of calcium on body weight and bone turnover markers in women participating in a weight loss program. Journal of the American College of Nutrition. 2007; 26(5):456-61. [DOI:10.1080/07315724.2007.10719636] [PMID]
- Babaei P, Damirchi A, Hoseini R. The interaction effects of aerobic exercise training and vitamin D supplementation on plasma lipid profiles and insulin resistance in ovariectomized rats. Journal of Exercise Nutrition & Biochemistry. 2015; 19(3):173. [PMID]
- Holecki M, Zahorska-Markiewicz B, Więcek A, Mizia-Stec K, Nieszporek T, Żak-Gołąb A. Influence of calcium and vitamin D supplementation on weight and fat loss in obese women. Obesity Facts. 2008; 1(5):274-9. [DOI:10.1159/000169831] [PMID] [PMCID]
- Asemi Z, Tabassi Z, Heidarzadeh Z, Khorammian H, Sabihi SS, Samimi M. Effect of calcium-vitamin D supplementation on metabolic profiles in pregnant women at risk for pre-eclampsia: A randomized placebo-controlled trial. Pakistan Journal of Biological Sciences: PJBS. 2012; 15(7):316-24. [DOI:10.3923/pjbs.2012.316.324] [PMID]
- Major GC, Alarie F, Doré J, Phouttama S, Tremblay A. Supplementation with calcium+ vitamin D enhances the beneficial effect of weight loss on plasma lipid and lipoprotein concentrations. The American Journal of Clinical Nutrition. 2007; 85(1):54-9. [PMID]
- Asemi Z, Foroozanfard F, Hashemi T, Bahmani F, Jamilian M, Esmaillzadeh A. Calcium plus vitamin D supplementation affects glucose metabolism and lipid concentrations in overweight and obese vitamin D deficient women with polycystic ovary syndrome. Clinical Nutrition. 2015; 34(4):586-92. [DOI:10.1016/j.clnu.2014.09.015] [PMID]
- Sung K, Bae S. Effects of a regular walking exercise program on behavioral and biochemical aspects in elderly people with type II diabetes. Nursing & Health Sciences. 2012; 14(4):438-45. [DOI:10.1111/j.1442-2018.2012.00690.x] [PMID]
- Greene NP, Martin SE, Crouse SF. Acute exercise and training alter blood lipid and lipoprotein profiles differently in overweight and obese men and women. Obesity. 2012; 20(8):1618-27. [DOI:10.1038/oby.2012.65] [PMID]
- Zhang F, Ye J, Zhu X, Wang L, Gao P, Shu G, et al. Anti-obesity effects of dietary calcium: The evidence and possible mechanisms. International Journal of Molecular Sciences. 2019; 20(12):3072. [DOI:10.3390/ijms20123072] [PMID] [PMCID]
- Orrenius S, Zhivotovsky B, Nicotera P. Regulation of cell death: The calcium-apoptosis link. Nature Reviews. Molecular Cell Biology. 2003; 4(7):552-65. [DOI:10.1038/nrm1150] [PMID]
- Sergeev I. 1, 25-Dihydroxyvitamin D3 induces Ca2+-mediated apoptosis in adipocytes via activation of calpain and caspase-12. Biochemical and biophysical Research Communications. 2009; 384(1):18-21. [DOI:10.1016/j.bbrc.2009.04.078] [PMID]
- Sergeev IN, Song Q. High vitamin D and calcium intakes reduce diet-induced obesity in mice by increasing adipose tissue apoptosis. Molecular Nutrition & Food Research. 2014; 58(6):1342-8. [DOI:10.1002/mnfr.201300503] [PMID]
- Ikeda K, Kang Q, Yoneshiro T, Camporez JP, Maki H, Homma M, et al. UCP1-independent signaling involving SERCA2b-mediated calcium cycling regulates beige fat thermogenesis and systemic glucose homeostasis. Nature Medicine. 2017; 23(12):1454-65. [DOI:10.1038/nm.4429] [PMID] [PMCID]
- Golic I, Velickovic K, Markelic M, Stancic A, Jankovic A, Vucetic M, et al. Calcium-induced alteration of mitochondrial morphology and mitochondrial-endoplasmic reticulum contacts in rat brown adipocytes. European Journal of Histochemistry. 2014; 58(3):2377. [DOI:10.4081/ejh.2014.2377] [PMID] [PMCID]
- Cho HJ, Kang HC, Choi SA, Ju YC, Lee HS, Park HJ. The possible role of Ca2+ on the activation of microsomal triglyceride transfer protein in rat hepatocytes. Biological and Pharmaceutical Bulletin. 2005; 28(8):1418-23. [DOI:10.1248/bpb.28.1418] [PMID]
- Kong J, Li YC. Molecular mechanism of 1, 25-dihydroxyvitamin D3 inhibition of adipogenesis in 3T3-L1 cells. American Journal of Physiology. Endocrinology and Metabolism. 2006; 290(5):E916-24. [DOI:10.1152/ajpendo.00410.2005] [PMID]
- Jones JR, Barrick C, Kim KA, Lindner J, Blondeau B, Fujimoto Y, et al. Deletion of PPARgamma in adipose tissues of mice protects against high fat diet-induced obesity and insulin resistance. Proceedings of the National Academy of Sciences of the United States of America. 2005; 102(17):6207-12. [DOI:10.1073/pnas.0306743102] [PMID] [PMCID]
Type of Study: Original |
Subject:
Basic Medical Science
Received: 2022/08/24 | Accepted: 2023/01/25 | Published: 2022/09/23
Received: 2022/08/24 | Accepted: 2023/01/25 | Published: 2022/09/23
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |